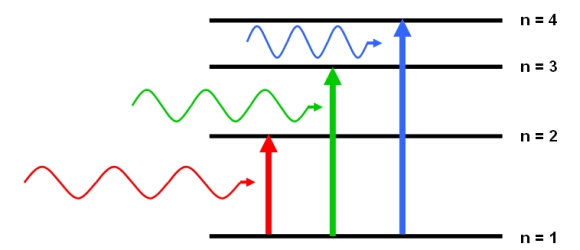
Absorption Line
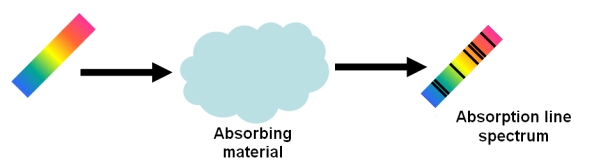
An absorption line will appear in a spectrum if an absorbing material is placed between a source and the observer. This material could be the outer layers of a star, a cloud of interstellar gas or a cloud of dust.
According to quantum mechanics, an atom, element or molecule can absorb photons with energies equal to the difference between two energy states.
Absorption lines are usually seen as dark lines, or lines of reduced intensity, on a continuous spectrum. This is seen in the spectra of stars, where gas (mostly hydrogen) in the outer layers of the star absorbs some of the light from the underlying thermal blackbody spectrum.
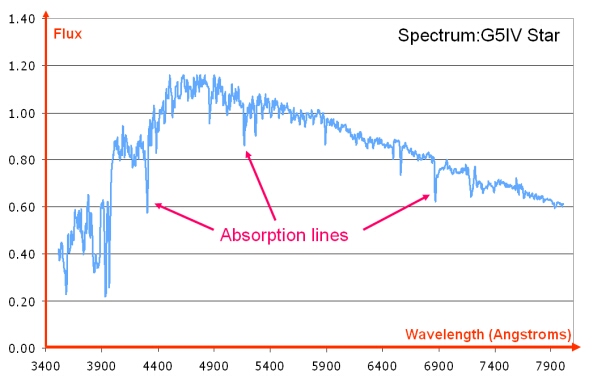
Dataset: VizieR catalogue III/219, Spectral Library of Galaxies, Clusters and Stars (Santos et al. 2002)
See also: spectral line.
Study Astronomy Online at Swinburne University
All material is © Swinburne University of Technology except where indicated.